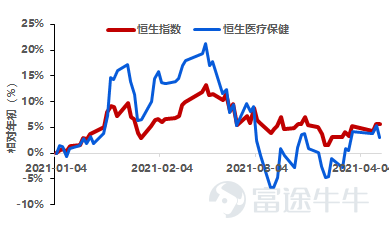
I. Industry Overview
The Hang Seng Healthcare Index fell by 1.13% this week, underperforming the Hang Seng Index by 0.3%. However, in terms of cumulative returns from the beginning of the year to date, the Hang Seng Healthcare Index significantly underperformed the Hang Seng Index by 3.04%.
Looking at the Futu Medical Health Index, the top three gainers this week were: Genscript Bio (+6.00%), Cansinobio-B (+5.82%), and Xintong Medical-B (+5.70%). The top three decliners this week were: Beigene (-12.07%), Junshi Bio (-7.52%), and Tigermed (-6.18%).
Analyzing by segment sectors, the medical equipment sector in the medical health industry performed well this week, while the performance of individual stocks in the innovative drugs and pharmaceutical outsourcing sectors varied significantly.

Second, viewpoints for this week
Chengdu Kanghong Pharmaceutical Group (002773.SZ), on April 9, announced the clinical progress of the global trial of Kangbixipu (PANDA Trial) that began in May 2018. The interim analysis of the unblinded data shows that the global public health events have significantly exceeded the company's expectations, with various countries' ongoing control measures leading to a large number of dropouts, loss to follow-up, and protocol deviations. The number of cases fully compliant with the trial dosing regimen has gradually decreased to less than 40% of the expected enrollment. The Scientific Steering Committee of the PANDA Trial believes that the trial did not meet its intended objectives and recommended the company to halt the trial. The company decided to stop the global PANDA Trial. Previously, Kangbixipu has had several years of sales in China, with high internationalization expectations. This decision to halt the global trial will have a significant impact on Kangbixipu's peak expectations, highlighting the challenges of global clinical advancement and affecting market expectations for the clinical progression of other international projects, significantly impacting the market sentiment of the innovative drugs sector.
Cansino Biologics Inc. - B (6185.HK; 688185.SH) is accelerating the commercial value realization of the COVID-19 vaccine. Earlier, Cansino's recombinant novel coronavirus vaccine (type 5 adenovirus vector) (AD5-NCOV) Kewei Sha™ has been approved in China, Mexico, Pakistan, and Hungary. On April 7th, the Chilean health regulatory agency approved emergency use of the Chinese Cansino vaccine, expected to arrive in Chile in May and June. Compared to similar products overseas, the Cansino COVID-19 vaccine only requires one dose, has minimal side effects, can be transported in regular cold chain, with a comprehensive protection rate of 65.7% and severe protection rate of 90.98%. The Cansino COVID-19 vaccine is more suitable for use in the current COVID-19 pandemic. Subsequent observations are needed on the capacity expansion progress of Cansino's Tianjin factory, with an annual production capacity expected to reach 0.2 billion to 0.25 billion doses of vaccines, to see if it can release capacity as scheduled, entering a phase of needing to compete in the market based on production capacity.
With the rapid progress of domestic innovative drugs, there are both shining moments of success and dark times of disappointment. Previously, Bio-Thera Solutions (688177.SH) terminated 3 new drug projects, and GL Pharm-B (1672.HK) quietly discontinued the marketed product Gonoevir® (Danorvir/ Longer-Acting Interferon and Ribavirin Gonoevir Treatment Regimen). Combined with the recent international clinical trial termination of Chengdu Kanghong Pharmaceutical Group (002773.SZ), the market may refocus on the research and development risks of new drugs, temporarily suppressing the valuation of innovation-driven pharmaceutical enterprises. In the long term, innovation undoubtedly remains the direction of the industry's development but is gradually transitioning from the FAST-FOLLOW ME-TOO stage to the FIRST-IN-CLASS stage.
IV. Risk alert
Product development failures or lower-than-expected market performance; regulatory approval and commercialization progress falling short of expectations.
Appendix
Performance and valuation of leading companies in various sub-sectors of the medical health sector this week.

Risk Disclaimer: The above content only represents the author's view. It does not represent any position or investment advice of Futu. Futu makes no representation or warranty.Read more
Comments
to post a comment
5