May 7, 2022,$GRAND PHARMA (00512.HK)$ It was announced that itsSIR-spheres® yttrium [90Y] microsphere injection, an innovative global product for treating malignant liver tumors, was officially put into use in China. Zhongda Hospital Affiliated to Southeast University, Tianjin Medical University Cancer Hospital, Hunan Provincial People's Hospital, and Xi'an Jiaotong University First Affiliated Hospital respectively performed and successfully completed precise interventional clinical procedures with yttrium [90Y] microsphere injections.This is the first batch of applications since the product was approved for marketing by the State Drug Administration this year. Its full implementation means that patients with malignant liver tumors in China have received new treatment methods, and the treatment of malignant liver tumors in China has entered a new “yttrium era”。
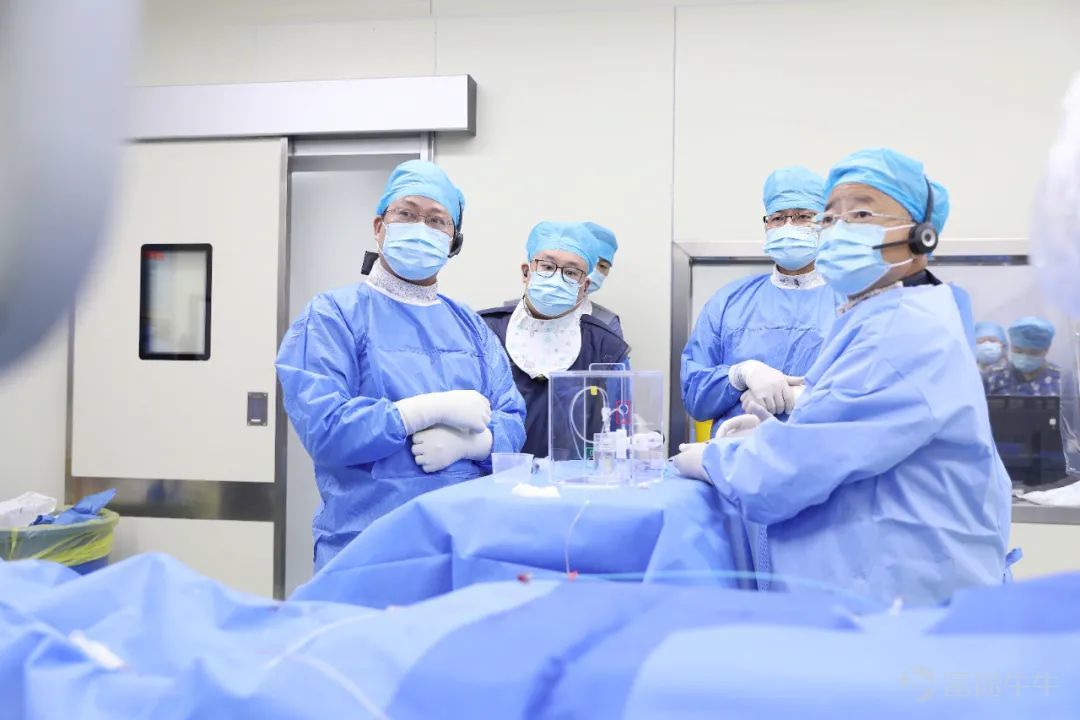
This was the first batch of precise interventional procedures with SiR-spheres® yttrium [90Y] microsphere injections performed at four hospitals, respectivelyProfessor Teng Gaojun, Academician of the Chinese Academy of Sciences, Director of Zhongda Hospital Affiliated to Southeast University, and Director of the Interventional Diagnosis and Treatment Center; Professor Lu Wei, Party Secretary of Tianjin Medical University Cancer Hospital and Director of the Liver Cancer Prevention and Treatment Research Center; Professor Xiang Hua, Executive Vice Director of the Hunan Provincial People's Hospital; and Professor Liu Qingguang, Deputy Director of the First Affiliated Hospital of Xi'an Jiaotong Universityimplementation.
On January 30, the National Drug Administration (NMPA) approved the marketing application for yttrium [90Y] microsphere injections. The indications are colorectal cancer liver metastases that have failed to be surgically resected after standard treatment. This is the first product in China for topical treatment of colorectal cancer liver metastases approved for marketing, indicating that a precise internal radiation therapy plan that meets international standards will be applied to patients with colorectal cancer liver metastases in China.

Professor Teng Gaojun of the Interventional Diagnosis and Treatment Center of Zhongda Hospital affiliated to Southeast University said, “This interventional internal radiation treatment is the first batch of use of yttrium [90Y] microsphere injections after being approved for marketing by the State Drug Administration. We are happy to see that innovative treatments with good clinical results in the field of international interventional therapy are currently being used by Chinese patients, and the successful implementation of surgery has given new hope to patients with colorectal cancer liver metastases. A large amount of data shows that yttrium [90Y] microsphere injections provide a good opportunity for reduction of the period and subsequent surgical conversion treatment.”

“Unlike traditional laparotomy, yttrium [90Y] microsphere injection is more like a micronuclear bomb with precise guidance. In the early stages, through precise positioning, yttrium [90Y] microsphere injections with reasonable radiation reach the inside of the tumor through the interventional procedure, and concentrate on rapidly killing tumor cells by releasing beta rays without affecting surrounding healthy tissue. Compared with traditional treatment models, yttrium [90Y] microsphere injection precision treatment technology has certain advantages and can bring more benefits to patients.” Professor Lu Wei, Secretary of the Party Committee of the Cancer Hospital of Tianjin Medical University and director of the Liver Cancer Prevention Research Center, gave an introduction.


Expert team leaders from the four hospitals said that accurate interventional procedures with yttrium [90Y] microsphere injections currently being carried out at the four hospitals went smoothly. During surgery at every hospital,The patient's vital signs are stable, and there are no adverse reactions. In order to ensure that patients can obtain good clinical treatment results, the hospital immediately carried out training on the product and technical capabilities after the product was approved for marketing by the State Drug Administration, and strictly evaluated and prepared hardware site wards, etc. At the same time, multidisciplinary participation also ensures that patients can receive comprehensive, accurate and personalized diagnosis and treatment throughout the entire diagnosis and treatment process before, during, and after surgery, laying a good foundation for the successful implementation of interventional surgery and subsequent transition.
AsThe US Food and Drug Administration (FDA) officially approved the first radioactive microsphere product officially used for colorectal liver metastasis. Sir-spheres® yttrium [90Y] microsphere injection has treated more than 120,000 patients in more than 50 countries and regions around the world, with no significant adverse effects. Its safety and efficacy have been fully verified, and recommended by mainstream diagnosis and treatment guidelines in the US, Europe, the UK and China. In September 2021, Dong Jiahong's team, an Academician of the Chinese Academy of Engineering, successfully carried out China's first precision intervention with licensed yttrium [90Y] microsphere injections in Hainan; the product was approved for marketing by the State Drug Administration in January 2022. Experts said that the first batch of accurate interventional procedures with yttrium [90Y] microsphere injections carried out by top expert teams from four key hospitals in China brought new hope to patients with malignant liver tumors in China. According to reports, many key hospitals in many regions of China will also successively carry out precise interventional procedures with yttrium [90Y] microsphere injections.
About liver cancer:
Liver cancer is known as the “king of cancer”. In February 2022, the National Cancer Center released the latest national cancer statistics, showing that China has 400,000 new cases of liver cancer every year, accounting for 47.6% of the world's new liver cancer cases. Most patients are in the middle to advanced stages of detection. Only 20% to 30% of liver cancer patients can be surgically removed, and the recurrence rate is still as high as 60% to 70% within 5 years. However, the Chinese Colorectal Cancer Liver Metastasis Diagnosis and Comprehensive Treatment Guidelines (2020) show that in China, liver metastases are an important cause of death directly in colorectal cancer patients. About 50% of colorectal cancer patients will have liver metastases, 80-90% of liver metastases cannot initially be radically removed, and the 5-year survival rate of patients with liver metastases that cannot be removed is less than 10%.
About Yttrium [90Y] Microsphere Injection:
In 2002, SIR-spheres® yttrium [90Y] microsphere injection was approved for marketing by the US FDA and the European Medicines Agency (EMA). Up to now, it is still the only “SIRT” product in the world for selective internal radiation therapy of liver metastases in colorectal cancer. In the 20 years since its launch, yttrium [90Y] microsphere injection has treated more than 120,000 patients worldwide, and its safety and efficacy have been widely recognized. It has been recommended by guidelines issued by the European Society of Medical Oncology (ESMO), the US National Comprehensive Cancer Network (NCCN), and the British National Health Care Professional Organization (NICE) to treat liver metastases in colorectal cancer. For patients with inoperable colorectal cancer liver metastases that failed standard treatment, post-marketing clinical study results showed that liver metastases can be effectively controlled after treatment with yttrium [90Y] microsphere injections, and the technical resectability rate of inoperable patients can also be increased, so that patients can regain the chance of surgery.
Yuanda Pharmaceutical and CDH Genetech Limited acquired Sirtex and introduced yttrium [90Y] microsphere injections in 2018. Yttrium [90Y] microsphere injection was approved for listing in China in August 2020 based on clinical trial data obtained abroad. In November of the same year, NMPA officially accepted the marketing license application (NDA) for yttrium [90Y] microsphere injection for colorectal cancer liver metastasis indications. In September 2021, Sirtex, with the assistance of Yuanda Pharmaceutical, successfully completed China's first authorized admission procedure for primary liver cancer with yttrium [90Y] microsphere injection, based on the policy of first testing medical devices marketed overseas in the Boao Lecheng International Tourism and Medical Pilot Zone of the Hainan Free Trade Port.
![Sir-spheres® yttrium [90Y] microsphere injection](https://nnqimage.futunn.com/8853341209696625684.jpg/big?imageMogr2/ignore-error/1/format/webp)
About Yuanda Pharmaceutical:
Yuanda Pharmaceutical Group Co., Ltd. (512.HK) is a technology-innovative international pharmaceutical enterprise. The group's core products cover several major business areas represented by anti-tumor, cardiovascular first aid preparations and high-end cardiovascular interventional medical devices, critical illness and anti-infection, respiratory and facial surgery, biohealth products and boutique APIs, and in the three innovative strategic directions of “precise cardiovascular and cerebrovascular intervention”, “intervention, nuclear drugs and immunotherapy against tumors” and “severe illness and anti-infection”, and has prepared 33 innovative global products distributed at different stages of pre-clinical marketing applications. The pipeline layout is reasonable, forming a good echelon effect.
The Group continues to accelerate the pace of global expansion. It has a sales network in more than 60 countries and regions, with customers in Asia, America and Europe. The Group has joined hands with major joint ventures Australian Sirtex, US OncoSec (NASDAQ: ONCS) and participating company Telix (ASX: TLX), and a number of overseas high-tech companies such as Cardionovum, ITM, and Belgium Etherna to lay out five technology R&D platforms and five R&D centers around the world to jointly promote the Group's international innovation layout.
Adhering to the development concept of “comprehensive advantages, innovation barriers and global expansion”, the Group adopts the “independent R&D and global expansion of two-wheel drive, global operation layout and dual cycle operation development” strategy, targets cutting-edge fields of scientific and technological innovation, continuously expands and implements new performance growth points from the perspective of internationalization and scientific and technological innovation. It is committed to building an internationally leading platform for precise interventional diagnosis and treatment, and has developed into a world-class nuclear medicine cancer diagnosis and treatment platform and a leading technology enterprise in the field of severe illness and infection prevention.
Forward-looking statements
This press release may include certain forward-looking statements. These statements are inherently quite risky and uncertain. When using “anticipation,” “belief,” “forecast,” “expectation,” “intention,” and other similar terms, everything relating to the Company is a forward-looking statement. The Company is under no obligation to continuously update these predictive statements.
These forward-looking statements are based on the Company's management's existing views, assumptions, expectations, estimates, predictions, and understandings of future matters at the time the statements are made. These statements are not a guarantee of future development and are subject to risk, uncertainty and other factors. Some are beyond our control and difficult to predict. Therefore, due to future changes and developments in our business, competitive environment, political, economic, legal and social circumstances, actual results may differ materially from the information contained in the forward-looking statements.
The Company, its directors and employee agents are not responsible for (a) any obligation to correct or update the forward-looking statements contained on this website; and (b) any liability arising from the failure or inaccuracy of any forward-looking statements.
Risk Disclaimer: The above content only represents the author's view. It does not represent any position or investment advice of Futu. Futu makes no representation or warranty.Read more
Comments
to post a comment
3