2026 IPO bonanza! Over 90% of new stocks rose on their debut!
Futu News May 13 report $IMPACT THERAP-B (07630.HK)$ Announced allocation results: priced at HKD 20.1 per share, issuing a total of 41.977 million shares, with each lot consisting of 200 shares, officially listed today.
In the last trading session, Impel Pharma's gray market closed up 60% at HKD 32.16 per share, with each lot containing 200 shares. Excluding fees, earnings per lot were HKD 2,412.
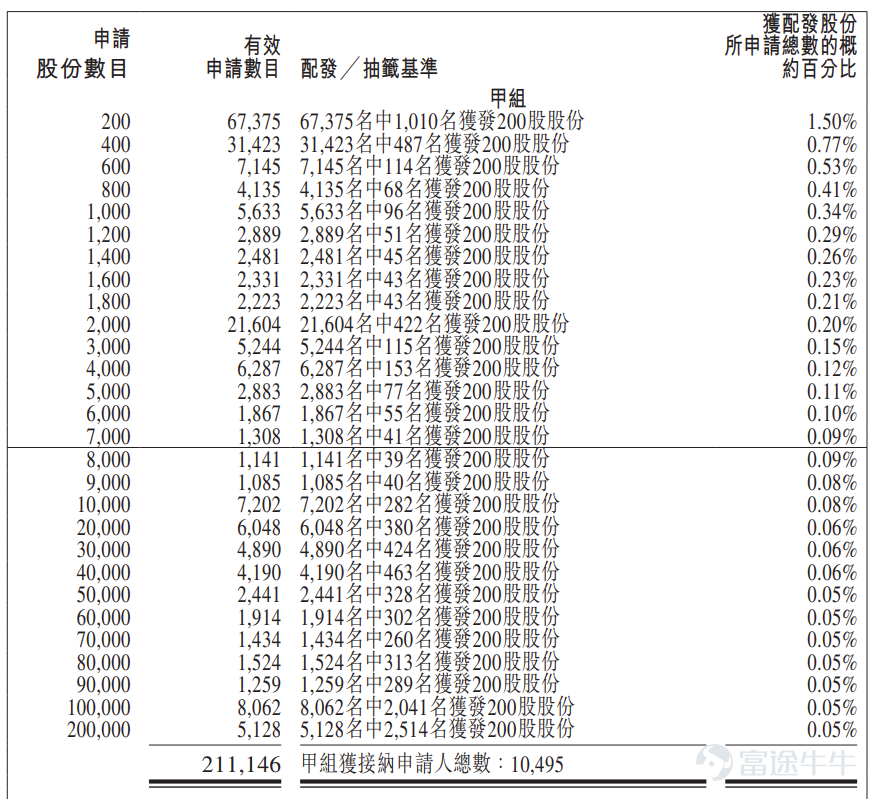
During the public offering phase, Impel Pharma received 2,282.4 times subscription. The final number of shares offered to the public was 4.1978 million, accounting for approximately 10% of the total shares issued. A total of about 221,800 valid applications were received, with approximately 18,774 accepted applications. The percentage of shares allocated per lot relative to the total applied shares was approximately 1.5%.
In addition, Impulse Therapeutics' international placement was subscribed 24.58 times, with the final number of international placement shares amounting to 37,779,200 shares, accounting for 90% of the total offering shares. Futu Information has compiled the relevant data in the table below:


Company Overview
Founded in 2009, Impaper Pharmaceuticals is a commercial-stage biotechnology company committed to advancing precision cancer therapies based on synthetic lethality mechanisms worldwide, developing innovative treatments to address unmet medical needs for cancer patients. Its core product, Senaparib, was approved in January 2025 for first-line maintenance treatment of ovarian cancer across the entire Chinese patient population, marking the start of its commercialization.
As of the latest practicable date, Impaper Pharmaceuticals has built a pipeline matrix comprising one commercialized product, four clinical-stage products, and seven pre-clinical products, covering small molecule inhibitors targeting key synthetic lethality pathways such as PARP1/2, PARP1, ATR, WEE1, and PKMYT1/WEE1. It is also exploring emerging therapies like ADCs and protein degraders. The company is one of only three enterprises globally with both a commercial-stage PARP1/2 inhibitor and a clinical-stage next-generation PARP1 selective inhibitor.
Financial Summary
According to the prospectus, InnoPar's revenue for 2024 and 2025 will be RMB 33.55 million and RMB 38.25 million respectively; gross profit for 2024 and 2025 will be RMB 31.99 million and RMB 36.68 million respectively, with corresponding gross margins of 95.4% and 95.9%; net losses for the periods in 2024 and 2025 will be RMB 255 million and RMB 296 million respectively.

Use of Proceeds
Regarding the use of proceeds, Impulse Therapeutics expects net proceeds from the global offering to be approximately HKD 760 million (based on an offer price of HKD 20.1). According to the prospectus, Impulse Therapeutics intends to allocate the proceeds from the global offering as follows:
Approximately 51% will be used to fund ongoing and planned clinical development, regulatory approval, and commercialization of the company’s core product, Senaparib; about 31% will be allocated to support the ongoing clinical development of the company’s key products IMP1734 and IMP9064; around 8% will go towards funding research and development activities for other pipeline assets, including IMP1707, IMP7068, IMP22, IMP25, IMP08, IMP13, and IMP10; approximately 8% will be used for the development and expansion of the company’s R&D platform and drug pipeline; about 2% will be utilized for working capital and other general corporate purposes.
The golden season for new stock subscriptions has arrived! Use Futu for new stock subscriptions—zero interest, zero handling fees, and zero cash subscription. Participants now have the chance to have their new stock subscription fees waived for a year.Come and experience it now >>

Editor/Joe
Risk Disclaimer: The above content only represents the author's view. It does not represent any position or investment advice of Futu. Futu makes no representation or warranty.Read more
Comments
to post a comment
8
7