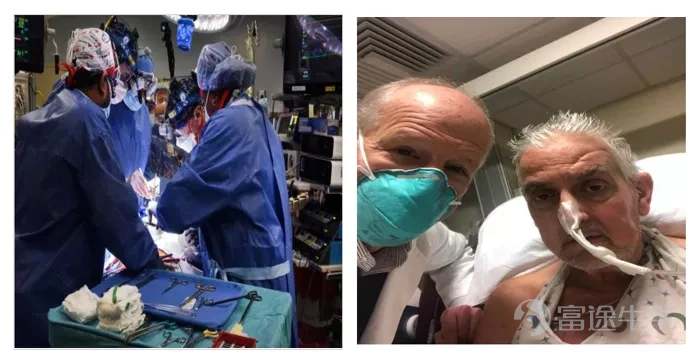
David Bennett, 57, has an advanced heart disease and his life is in jeopardy. After reviewing his medical records, Bennett was deemed ineligible for a traditional heart transplant or artificial heart pump.Pig heartIt's “the only option currently available.”
“Either die or have a transplant. I want to live. I know it was a shot in the dark, but it was my last resort,” Bennett said before surgery.
In order to advance experimental surgery,University of MarylandIt received emergency authorization from the FDA on New Year's Eve through its compassionate use program.
David Bennett is lucky!After nearly eight hours, doctors at the University of Maryland School of Medicine successfully transplanted the heart of a gene-edited pig for the first timeDavid BennettIn the body。After 3 days of surgery, David Bennett is still in good health, with no significant organ rejection (Figure 1).
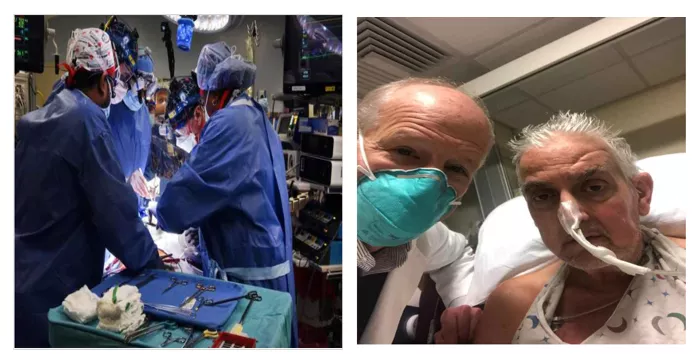
This is the first time in the world that a human underwent a pig heart transplant,This is clearly a pioneering operation that, if proven successful, will provide hope for tens of thousands of patients with organ failure.
Pigs: the ideal organ source for human xenografts
An important option for organ regeneration engineering is to use live donations from other species as a source. The advantages of this approach are enormous. Naturally living organs are fully functional, but cross-species immune responses also present challenges. Genetic engineering is necessary to overcome this limitation. The long-known hyperacute immune response to animal organs is largely eliminated by knocking out genes encoding the synthesis of cell-surface carbohydrate heterologous antigens. The most important gene is GGTG1, which codes for alpha-1, 3-galactosidase. Two other genes (CMAH and B4GALNT2, which encode proteins that produce N-hydroxyneuraminic acid) and the SD (A) heterologous antigen have also been studied in gene knockout models to reduce cross-reactions. Other challenges associated with xenotransplantation include the risk of zoonotic diseases associated with the animal species that provided the organs, and the patient's own reluctance to receive organs of animal origin.
Pigs are generally considered an ideal source of organs for human xenotransplantation. First, pigs and human organs are similar in size and shape, and are easy to obtain. Both inbred and long-bred lines have been used in preclinical research. Furthermore, pigs can be cloned via somatic cell nuclear transfer technology, and their genomes can now be easily edited with nucleases such as zinc finger nucleases, transcription-activator-like effector nucleases (TALENS), CRISPR-Cas9). Finally, since pigs are more closely related to humans than non-human primates, they are much less likely than non-human primates to spread pathogens through organ transplants.
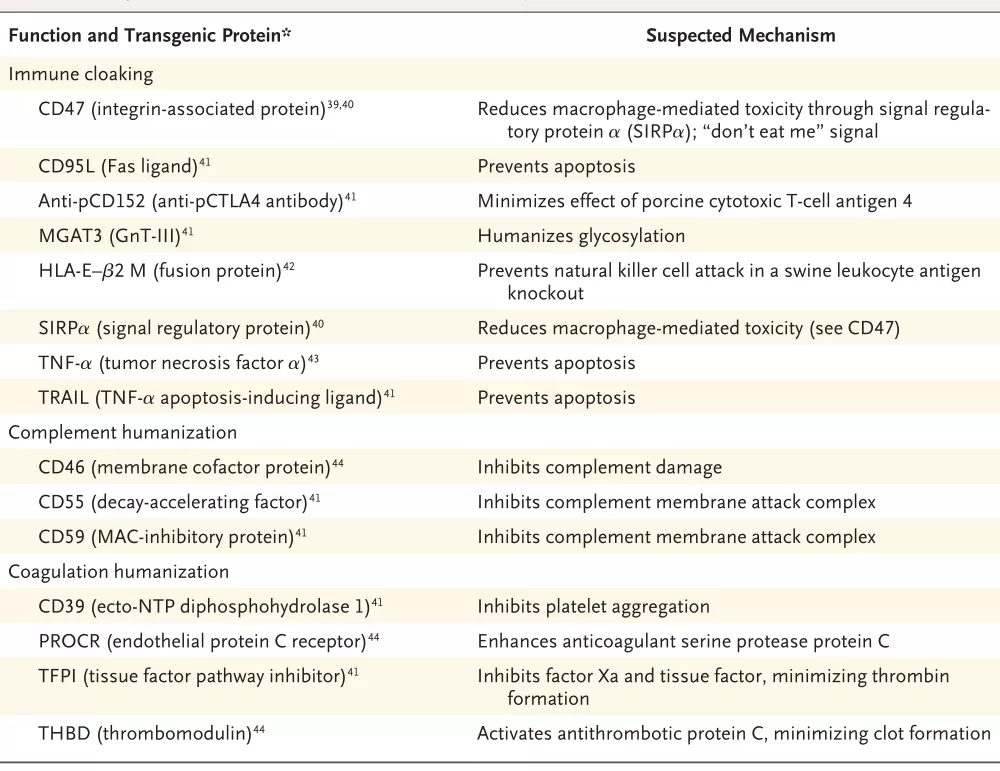
Whole-organ xenografts were performed in the early 20th century, and news about them is mixed. It is now recognized that most organ transplants quickly fail due to hyperacute rejection of glycosylated proteins (such as galactose-alpha-1, 3-galactose). According to reports, knocking out the GGTG1 gene responsible for primary glycosylation activity in donor pigs through genetic engineering can greatly reduce hyperacute rejection. However, when this tissue was transplanted into non-human primates, it was still rejected within a few weeks. Subsequent researchers carried out a series of studies to determine human genes that mainly encode the CD (differentiation cluster) membrane protein family (Figure 2), which can be expressed as genetically modified proteins, fusion proteins, or even antibodies against porcine protein to further reduce the human immune response against pigs. Over the past decade, several such genes have been tested, and the proteins they code for are mainly divided into three categories: immunocclusion, that is, specific transgenic proteins interfere with the killing of NK or T cells, and complement humanization and coagulation humanization, that is, use human complement or human coagulation pathways to replace or supplement corresponding pathways in pigs. These interventions greatly improved the chances of successful xenotransplantation into non-human primates, and organ rejection was substantially improved.
Revolutionary progress and future of xenotransplantation
There have been two notable developments in xenotransplantation over the past few years. The first concerns the safety of transmission of porcine endogenous retroviral sequences (PERVs) to transformed human tissue culture cells. To completely eliminate this risk, CRISPR-Cas9 technology was recently used to eliminate all 62 copies of PERV present in the pig genome in PK15 pig kidney cells and fibroblasts. The second group, which had revolutionary progress, produced organ-specific interspecific chimeras (sometimes called “exogenesis”), which opened the door to the possibility of successfully producing organs containing complete human cells in chimeric animals (Figure 3).

Huge advancements may make “traditional” xenotransplantation not only viable, but practical. Studies have been conducted on so-called three-gene knockout pigs (that is, GGTA1, CMAH, and B4GALNT2 gene knockout). These pigs lack carbohydrate heterologous antigens on the surface of pig cells, and these antigens are usually recognized by normal human serum and peripheral blood monocytes. According to reports, pigs knocked out of these three genes are healthy. In vitro studies have shown that a significant proportion of the human population lacks antibody responsiveness to renal microvascular epithelial cells from these animals. Furthermore, the degree of cross-reaction observed in some patient samples is similar to that observed in some human allogeneic transplant recipients, which suggests that for such patients, xenograft rejection from such animals may be controlled by using existing immunosuppressive protocols. Human cell subsets that maintain an immune response to trigene knockout cells appear to recognize porcine leukocyte antigen (SLA) class I (equivalent to porcine MHCI). In a subsequent study, the researchers assessed the effect of porcine SLA gene deletion on serum immunoreactivity analysis in patients on the transplant waiting list. The results showed that this SLA deficiency may increase the proportion of patients benefiting.
The second method to minimize immune damage to pig organs after transplantation in humans is to engineer complement components and pathways. One way to mitigate this damage is to humanize porcine surface antigens by expressing one or more human genetically modified proteins that downregulate human complement activity, and then “mask” them so they cannot be recognized by the human immune system. Due to the co-evolution of human complement regulatory proteins and human complement pathways, human protein is expected to modulate the effects of complement more effectively than porcine protein. These proteins include CD55 (complement decay acceleration factor), CD59 (membrane attack complex inhibitory protein), and CD46 (complement regulator). The gene encoding tumor necrosis factor alpha is also thought to play an important role in controlling acute vascular rejection in pig transplants, and the CD47 signaling protein — which sends a “don't eat me” message — is also particularly beneficial in promoting the receptivity of pig tissue.
Hozain and others recently reported another cross-species innovation in the field of transplantation, which revitalizes human lungs initially thought to be too “damaged” to be transplanted by connecting human lungs to pig circulatory systems. This extracorporeal circulation restores damaged lungs to a sufficiently healthy state, which may be suitable for lung transplantation. This strategy can greatly increase human lung availability.
Editor's summary
Organ transplantation is the most complete option in regenerative medicine, providing a self-contained, allogeneic, or potentially heterologous alternative for complete physical and biological repair. Advances in immune and genomic engineering (or editing) are laying the foundation for new treatments to accelerate tissue and organ xenotransplantation. Xenotransplantation has made groundbreaking and revolutionary advances in the past few years. We look forward to David Bennett being reborn, and we also expect tens of thousands of people with organ failure like David Bennett to get rid of the pain of illness as soon as possible.
Source: Biopharmaceutical Editor
Risk Disclaimer: The above content only represents the author's view. It does not represent any position or investment advice of Futu. Futu makes no representation or warranty.Read more
Comments
to post a comment
1